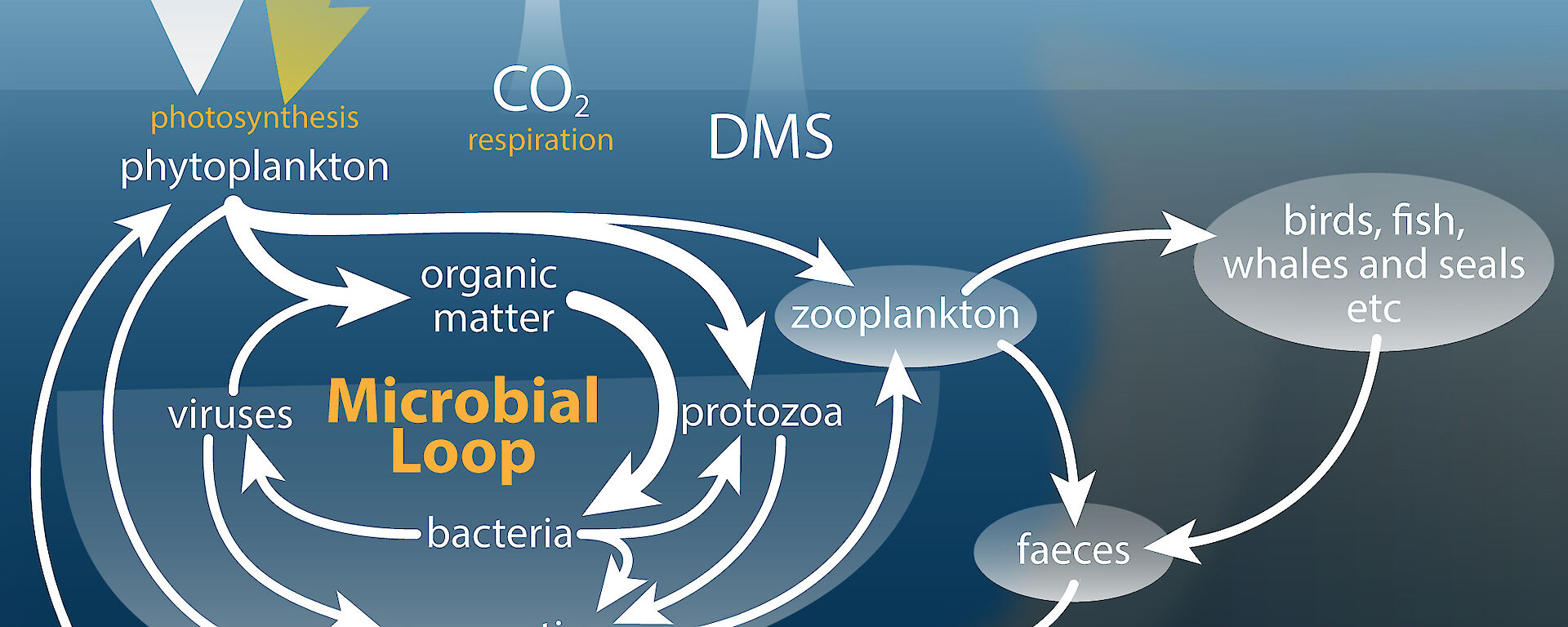
Antarctic marine microbial communities — bacteria, phytoplankton and protozoa — are the base of the food web and, directly or indirectly, support all life in the Southern Ocean. However, the concentration of carbon dioxide (CO2) predicted in seawater by the end of this century may dramatically change the composition of these communities, with consequences for iconic Antarctic wildlife and climate.
To investigate the effects of increasing CO2 on Antarctic marine microbes, we installed six 650 litre ‘minicosms’ (incubation tanks) in a shipping container on the shore at Davis station in 2008–09. The tanks were filled with seawater, which was filtered to remove any organisms larger than 200 µm (0.2mm).
Different volumes of CO2-saturated, sterile seawater were then added to each of the tanks to achieve CO2 concentrations of 84, 643, 1281, 1848, 1942 and 2423 ppm (parts per million). These corresponded to approximately 600 ppm increments from ambient Antarctic concentrations in summer, to the maximum seawater CO2 concentration predicted for 2300.
Antarctic marine microbes naturally experience a wide seasonal range in CO2 concentrations. In summer, intense phytoplankton blooms consume the CO2, reducing its concentration from the current global average of approximately 386 ppm* to only 100 ppm. In winter the CO2 concentration can increase to about 450 ppm, due to upwelling of CO2-rich deep water, little or no light for photosynthesis, and the presence of sea ice which limits gas exchange between the atmosphere and ocean.
Perhaps unsurprisingly, our experiments using natural communities of Antarctic marine microbes at Davis showed that they can tolerate the large seasonal fluctuations they experience in nature, as there was little effect at 84 and 643 ppm CO2 (0.2 and 1.7 times the current atmospheric levels). However, there were dramatic changes in the species composition and size of microbial cells at higher CO2 concentrations.
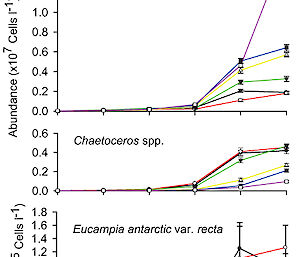
When we exposed the microbial community to 1281 ppm CO2 and above (≥ 3.3 times current CO2 levels) we saw a rapid shift in the size of phytoplankton. The abundance of large phytoplankton above about 20 µm (0.02mm) in size greatly declined, while there was an increase in the number of small species less than 5 µm.
The decrease in the size of phytoplankton coincided with a decline in the amount of phytoplankton (chlorophyll a and carbon). Thus, at CO2 concentrations predicted for around the end of this century (near 1000 ppm), we saw a threshold above which productivity drops and cells get smaller.
At concentrations ≥ 1281 ppm CO2 we also saw the abundance of bacteria increase, but the growth of protozoa, which graze on bacteria and small algae, slowed.
These results suggest that protozoan grazing controlled the abundance of small cells at low CO2. At ≥ 1281 ppm, inhibition of protozoan growth allowed small cells to escape this control and their abundances increased. So, the effects of CO2 concentration are seen as changes in the relative abundance of species within a single level of the food web (e.g. phytoplankton), but can be driven by CO2-induced changes in the interactions among organisms across trophic levels (e.g. between phytoplankton and protozoa).
The decline of larger phytoplankton under higher CO2 levels could have a dramatic impact on the entire Antarctic ecosystem towards the end of this century. Krill are one of the key grazers of phytoplankton, but can only eat cells bigger than 6 µm. A decline in suitable food would reduce krill abundance, with knock-on effects for the many other species that eat krill, including seabirds, seals and whales.
But the consequences don’t end there; the increased abundance of small phytoplankton cells could affect the global carbon cycle. Under normal conditions phytoplankton incorporate CO2 into their cells during photosynthesis. Some cells, often the large ones, sink to the deep ocean, carrying this carbon with them. This process mediates the concentration of CO2 in the Earth’s atmosphere. The changes we observed, however, would act as a positive feedback on global warming. Small phytoplankton cells sink more slowly and are commonly consumed in near-surface waters by protozoa and bacteria. Much of the carbon stored in these cells is respired by the protozoa and bacteria, and released back to the atmosphere as CO2.
If our experiments are to help us understand the consequences of rising CO2 to Southern Ocean ecosystems, we must be confident that the contents of the minicosms mimic what is happening in the natural environment. We plan to repeat our experiments at Davis in the 2013–14 summer to determine whether the responses are consistent among years. And we need to refine our estimate of the threshold CO2 concentration at which the change in the microbial community occurs.
We also need to better understand the possible mechanisms causing these responses. Iron, for example, is a limiting micronutrient over much of the Southern Ocean. When you change the chemical quality of seawater you change the dynamics of its trace metal chemistry — so there might be plenty of iron in the water but it may be in a form that is unavailable to phytoplankton. CO2 might also affect the process of photosynthesis in some species, and we don’t yet understand the mechanism by which CO2 is inhibiting the small protozoa.
So much remains to be learned quickly if we are to predict future marine microbial community responses, inform policy development, and avoid possible catastrophic effects on the Antarctic ecosystem.
Andrew Davidson
Marine microbial ecologist,
Australian Antarctic Division
* At the time of the experiments in 2008–09 the atmospheric concentration of CO2 was 386 ppm. In 2013 it is 394 ppm.