Dr Kawaguchi’s praise for the Australian Antarctic Division’s krill aquarium has proved well founded. Since his appointment, Dr Kawaguchi and his team have significantly advanced scientific knowledge about the biology and behaviour of Antarctic krill (Euphausia superba), and contributed to a precautionary approach for the management of the Southern Ocean krill fishery through the Commission for the Conservation of Antarctic Marine Living Resources (CCAMLR).
As one of only two krill aquariums in the world — the other is a public aquarium in Japan — it supports a large number of international collaborations, including with Germany, United Kingdom, United States, China, Japan, Poland and Korea.
Critical to the aquarium’s success has been the ability to ‘close the life cycle’ of the crustacean — raising krill from egg to adult, through 12 larval stages — and maintaining them in conditions that support natural behaviour. These captive-bred krill supplement the 20 000 wild krill caught each year and enable experiments on specific life stages or animals of known age.
So how has the Australian aquarium advanced the collective understanding of krill?
Reproduction
To breed krill in captivity, scientists had to first develop a way of maintaining wild krill in healthy condition. The breakthrough came with a mechanism that enabled the aquarium water to be filtered at 20°C rather than 0°C. This increased the rate of gas exchange (adding oxygen and removing carbon dioxide) and the speed with which nutrients and waste products could be removed through a biologically active filter.
“These two advantages permitted the maintenance of more krill, and importantly, each individual could be fed a large amount of food, re-creating natural levels of food intake during krill maturation,” Dr Kawaguchi said.
With these systems in place and krill successfully spawning, the team was able to build on the work of the Port of Nagoya Aquarium in Japan and extend their understanding of the reproductive process and how they might be able to manipulate it.
In Antarctica, krill experience four months of darkness over winter, between June and September. During this time they actually shrink in size and lose their mature reproductive characteristics — a process known as regression. As spring approaches and the day length and food supply increases, they begin to grow and become sexually mature again, before mating begins in December and continues until about February.
In 2003, Japanese research showed that abrupt changes in the daily cycle of light and dark could trigger maturation and spawning. Then in 2009, the Antarctic Division team showed that it was the timing of the period of complete darkness, rather than the changing light, which alters the reproductive cycle in krill.
“We found that the annual cycle of krill maturation and reproduction can be altered by a period of darkness immediately after spawning,” Dr Kawaguchi said.
“We were able to reset the animals’ internal clocks so that they became sexually mature three months earlier than if they were exposed to a natural Antarctic life cycle.”
The discovery allowed scientists to control when different groups of krill spawn, so that they have access to all phases of the life cycle all year round, for experiments and breeding.
Scientists’ understanding of the sex lives of krill took another leap forward in 2011 when, in a world first, video footage of krill mating in the wild was captured at 507 m depth (Australian Antarctic Magazine 20: 11, 2011). The sighting provided the first evidence that mating can take place near the sea floor.
“This research challenges the assumption that most krill live within the top 200 m of the ocean and suggests that deeper layers are significant habitat for krill,” Dr Kawaguchi said.
Behaviour
Besides reproduction, another benchmark of healthy krill is the demonstration of natural behaviour.
In 2005 the Australian Antarctic Division aquarium managed to achieve repeated krill schooling behaviour in captivity. This means the team can now study the animals’ natural behaviour and response to various stimuli, and collect more accurate information on their growth and metabolism.
“Krill are very social animals, but in captivity they tend to behave individually, rather than as a group, so the information you can gather from them is limited,” aquarium manager and marine scientist, Mr Rob King, said.
“Normal behaviour, including schooling, means they should be physiologically normal, rather than stressed.”
Observations and measurements made on schooling krill are important because the information is used in modelling for fisheries research. It also contributes to scientists’ understanding of the interactions between krill, their predators, and the fishery, to improve krill fishery management under CCAMLR.
The team is now taking advantage of this success to conduct behavioural experiments using the latest camera and acoustic technologies.
Sound profile
The aquarium team has observed that when krill are feeding they swim around randomly, but when schooling, they swim faster, with their bodies close to horizontal and all pointing in the same direction. This information is important when measuring the abundance of krill at sea using acoustic ‘echosounders’. These instruments send pulses of sound at different frequencies into the water and record the echo strength as the sound waves bounce off objects. However, as the echo-sounder is sensitive to the body orientation of the krill, the echo strength may be different depending on what the krill are doing. If they are swimming horizontally, for example, the echo strength will be far stronger than when the krill are more vertical.
“When we use an echo sounder to observe a krill swarm on a ship like the Aurora Australis, we want to be able to convert the signals received into the number of krill that make up the swarm,” biological oceanographer Dr Martin Cox said.
“Modern echosounders use a range of frequencies and we don’t know what krill look like at these different frequencies and when they’re oriented differently to the source of the sound. To find out, we need to understand the reflection profile of an individual krill.”
To do this, Dr Cox has developed a new aquarium experiment.
“We’re taking individual krill of known lengths from the Antarctic Division’s aquarium, putting them in a purpose-built tank and measuring the sound that bounces off them across a range of frequencies. At the same time we’ll monitor their position with underwater cameras,” Dr Cox said.
Based on each individual krill’s sound profile, scientists will be able to scale up to determine the number of krill in a swarm. Dr Cox said the experimental results will also allow scientists to interpret data collected by krill fishing vessels.
“There are a lot of fishing boats that are using advanced echo sounders, but they often use them on a different frequency to the ones we have traditionally used and we don’t have data on how krill reflect sound on these frequencies,” he said.
“This work will ultimately enable more accurate measurements of krill density in the field, which is important for developing sustainable catch limits.”
Lifespan
The question of how long krill live has been occupying scientists for decades, and was a focus of the ‘father of modern krill research’, Dr Tom Ikeda, when he joined the Australian Antarctic Division from Japan in 1982. During his five-year tenure Dr Ikeda undertook pioneering work on krill longevity that showed they lived longer than the conventional wisdom of 3–4 years, and more like 5–6 years in the wild. He also demonstrated that krill shrink in the absence of food.
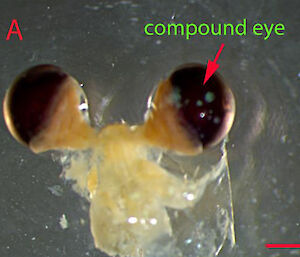
As a result of this quirk of nature, wild krill have proved difficult to age, adding a layer of complexity to krill fishery management. However, in early 2017, Dr Kawaguchi and collaborators from Canada, United States and Japan, showed that krill grow annual bands in their eyestalks, much like growth rings in trees, and that these correlate directly with their age.
“There’s almost no size difference in krill beyond two years of age, and their regular moult means they can actually shrink in size, depending on the time of year and food availability,” Dr Kawaguchi said.
“Our research has shown that we can look at a longitudinal section of the eyestalk to identify the light and dark growth bands and count exactly how many years the specimen has been alive.”
The age-based assessment methods will now provide information on stock structure to assist with determining catch limits and management options for the krill fishery through CCAMLR.
Growth rates
To accommodate krills’ ability to shrink and grow in size, Antarctic Division scientists have also recently developed a new model of krill productivity (growth and reproduction) to assist in sustainable krill fishery management.
The ‘energetics and moult cycle model’ couples the energy required from food for metabolism, growth and reproduction, and the constraints on growth during moulting — when krill shed and regrow their exoskeleton. The model also accounts for the effects of environmental change (temperature and food) on krill growth and reproduction.
“The interaction between food, temperature and the moult cycle is important in determining growth and reproduction in Antarctic krill,” Dr Kawaguchi said.
“The moult cycle physically limits the rates of shrinkage and growth in krill, and they can gain and lose weight within their exoskeleton. The extreme seasonality of the Antarctic means that reproduction is also dependent on food and whether the animals’ reproductive organs are active after winter.
“Our new model is sensitive to variability in regional, inter- and intra-annual food and temperature, and allows projections of krill growth in future environments.”
Ocean acidification
As more carbon dioxide (CO2) is pumped into the Earth’s atmosphere through human activities, oceans around the world, and particularly the Southern Ocean, absorb large amounts of the gas. This sets up chemical reactions in seawater that lead to a gradual increase in the oceans’ acidity.
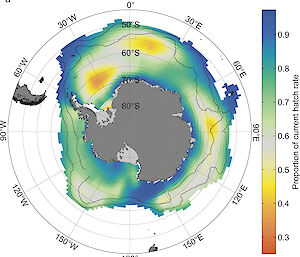
For more than 10 years, Dr Kawaguchi and Mr King have been studying the effect of ocean acidification and other climate-related stressors on Antarctic krill reproduction and development. Their experiments have shown that if carbon emissions and ocean acidification continue unabated, krill reproduction and development could be significantly reduced by 2100 and the entire Southern Ocean population could collapse by 2300. Current atmospheric CO2 levels are about 410 parts per million (ppm).
“We found egg hatch rates significantly decreased at CO2 levels of or above 1250 ppm with almost no hatching at 1750 and 2000 ppm,” Dr Kawaguchi said.
“We also found that embryonic development was significantly impaired if eggs are exposed to 1750 ppm during the first three days following spawning.”
Risk maps developed by the team show that much of the present habitat for Antarctic krill will experience damagingly high CO2 levels of above 1000 ppm by 2100 if emissions continue unchecked (Australian Antarctic Magazine 25: 4–5, 2013).
The results of this work will be incorporated into models of the dynamics of krill populations to assess the regional impacts of ocean acidification on the reproductive success of these populations. This information will be used to inform ecosystem and krill fishery management models under different carbon emission scenarios.
Genetic catalogue
Genetic technologies enable scientists to investigate a range of ecological questions about krill and other Antarctic animals, such as diet, population structure, and response to environmental stressors.
Molecular biologist, Dr Bruce Deagle, has been using krill DNA to determine whether different krill populations exist around Antarctica, and whether there is a genetic basis behind the sensitivity of krill to ocean acidification (Australian Antarctic Magazine 29: 6–7, 2015).
To look at population structure, Dr Deagle and his colleagues sequenced genetic markers across the krill genome, from krill collected from five areas of the Southern Ocean — covering the Atlantic, Pacific and Indian Ocean sectors. However, the team was unable to identify distinct DNA ‘fingerprints’ that would suggest the krill were from different populations.
“This supports the idea that there is a continuous flux of krill between regions carried by the Antarctic Circumpolar Current, and that genes are mixed before any regional genetic structuring develops,” Dr Deagle said.
To examine the genetic response of krill to ocean acidification, Dr Deagle looked at how the expression of different genes change under different CO2 concentrations. To do this he had to first create a detailed catalogue of krill genes (a ‘transcriptome’) through DNA sequencing.
While about 60 000 genes were already known from research by others, Dr Deagle’s team, and colleagues from the University of Padova in Italy, identified at least another 80 000 (the transcriptome database is available online).
“This catalogue is being used in several other projects at the Antarctic Division and internationally, looking at everything from how krill synchronise with their seasonal environment, to the mechanisms of krill sex determination,” Dr Deagle said.
“We have now produced a web-accessible database to make this transcriptome information widely available.”
By looking at the effect of CO2 exposure on the expression of these genes, Dr Deagle has been able to identify a number that appear to be affected. These genes and their functions are still being investigated in krill aquarium experiments and by collaborators from the Alfred Wegener Institute in Germany.
Fisheries management
Another aspect of the team’s work involves analysing information on the size of krill catches and fishing fleet behaviour (where ships catch krill and for how long), which is voluntarily provided to CCAMLR by most member countries.
“You can’t put scientific knowledge in place without understanding the commercial nature of the fishery,” Dr Kawaguchi said.
“It is also important to understand fleet behaviour in relation to the krill predators’ foraging ground. CCAMLR allows rational use of the resource, but we need to make sure that as the fishery develops it does not have any irreversible effect on the ecosystem.”
Some krill boats have recently begun using new equipment that allows larger volumes of krill to be caught and processed efficiently. This will change the dynamics of how fishing fleets interact with the resource, and the impact on krill predators — something Dr Kawaguchi and his colleagues will monitor with interest.
Catching krill for the future
Krill is collected from the wild for the Antarctic Division aquarium each year, using nets and a range of unique traps and pumps developed by Mr King and the Antarctic Division’s Science Technical Support team.
Among the novel technologies tested were deep sea light traps and cameras, to see if krill were feeding at the sea floor, up to 5800 m below, and to bring samples to the surface.
In 2012 Mr King trialled a modified fish pump to suck 400 litres per minute of water from beneath the sea ice and run it across a filter table onboard the ship (Australian Antarctic Magazine 23: 9, 2012). While it sounds like a good way to turn krill to mush, the pump was much gentler on the animals than traditional zooplankton nets, and more than 95% of the krill captured were in good condition and survived.
Further trials and improvements since then have shown the technique is a viable way to catch krill and other fragile marine creatures in good condition for experiments. The work has resulted in the inclusion of a specially designed ‘wet-well sampling space’ in Australia’s new icebreaker, currently under construction (Australian Antarctic Magazine 31: 7–8, 2016).
“In the wet well, water will gravity feed to large viewing tanks and filter tables,” Mr King said.
“Adult and larval krill will end up at the end of these tables where we can collect them and transfer them to an aquarium, while more fragile organisms, such as jellyfish, can be collected from the viewing tanks.
“There are many fragile species that we know little about because only divers can collect them in perfect condition. The wet well should allow us to collect these creatures alive, so that, for the first time, we’ll be able to study their growth rates, physiology, reproduction and behaviour.”
Krill research at the Australian Antarctic Division has come a long way in 35 years, and the krill team is well placed to continue advancing research into this keystone Southern Ocean species into the future.
Wendy Pyper
Australian Antarctic Division