Australian scientists will create a ‘future ocean’ under the sea ice off Casey this summer, using four underwater chambers to measure the impact of ocean acidification on seafloor (‘benthic’) communities.
Ocean acidification is caused by increasing amounts of atmospheric carbon dioxide (CO2) dissolving into seawater. This causes the pH of the seawater to drop and become more acidic, which affects the ability of some marine organisms, including corals, bivalves and some phytoplankton, to form shells and other hard structures. It also disrupts other important physiological processes (see fact box below).
The four semi-enclosed chambers will be deployed on the seafloor, 10 to 20m beneath the Antarctic sea ice off Casey station, between November 2014 and March 2015.
During the Antarctic ‘Free Ocean Carbon Enrichment’ (antFOCE) experiment, a team of scientific divers, technicians and engineers, will increase CO2 concentrations in the water in two of the chambers. This will decrease the pH of the water (by 0.4 pH units), without changing light or nutrient concentrations. A further two chambers will be used as ‘controls’ to track natural fluctuations in pH in the surrounding water. This will allow the team to compare the response of benthic communities exposed to current seawater pH levels, and the more acidic pH levels predicted under future CO2 emission scenarios.
A variety of observations and measurements will be made to allow the team to determine how benthic marine habitats respond to decreased seawater pH. These include:
- changes in benthic invertebrate community biodiversity and composition in sediments and on hard substrata;
- seawater carbonate chemistry;
- nutrient cycling in sediment;
- changes in communities of bacteria, diatoms (single celled marine plants) and sediment ‘meiofauna’ (very tiny critters);
- ‘bioturbation’ — how the actions of animals mixes the sediment; and
- ‘primary production’ — the growth of marine plants such as diatoms.
The research will assist governments, scientists, modellers and society to understand the emerging impact of ocean acidification on marine ecosystems and to ensure that the most relevant information underpins decisions to manage the threat.
Pumping gas
The underwater chambers used in the Antarctic Free Ocean CO2 Enrichment experiment are adapted from a prototype developed by the Monterey Bay Aquarium Research Institute in California. To date, this technology has been deployed in experiments run at sites in deep and shallow temperate waters and in shallow tropical and sub-tropical waters. As a result, the Australian Antarctic Division team has had to adapt the design and deployment of the chambers for the harsh Antarctic conditions.
Earlier this year the team spent several weeks testing the chambers in cold water off the coast of Tasmania, to practice deploying them and ironing out as many glitches in their operation as possible.
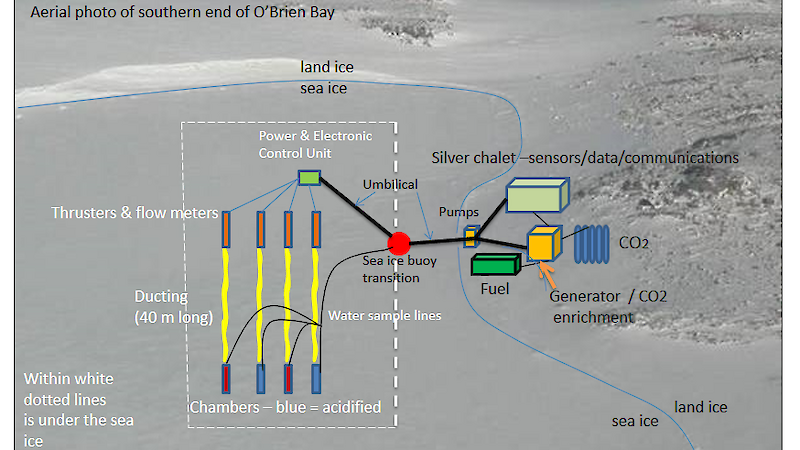
The Antarctic chambers are each coffee table-sized acrylic chambers (2m x 0.5m x 0.5m) anchored to the sea floor and sufficiently robust enough to withstand the −1.8°C water temperature. A series of pipes, linked to pumps, draw seawater to the surface, where it is enriched with CO2 , before being pumped back down under the sea ice and into the two experimental chambers. This provides a constant flow of CO2 -enriched, low pH seawater, through the two chambers.
Sample water drawn up from the experimental and control chambers passes through a series of pH, conductivity, temperature and oxygen sensors in the ‘Silver Chalet’ — a small shore-based control centre above the sea ice (see diagram). Underwater flow meters, thrusters, pH and temperature sensors are all powered from this surface hub.
Data collected from loggers in the Silver Chalet is wirelessly relayed back to station, allowing the technical team to monitor the experiment’s progress when no-one is on site. Last but not least, time-lapse cameras mounted on top of the chambers record how the structure of surface sediments in the chambers is changing over time.
Sea ice and ‘fast’ ice (attached to land) pose one of the biggest challenges to the success of the project. As there’s a reasonable tidal range at Casey, for the duration of the experiment there will be a few metres of sea ice grinding against the shore, bringing one tonne of force per cubic metre of sea ice.
To avoid this problematic zone, the project’s technical team has developed an original solution. Water pipes and power and data communication cables will be bundled together into an ‘umbilical’. This umbilical will extend from the shore-based control units and pass over the tide cracks, where sea ice meets and rubs up against the fast ice. They will then run under the sea ice through a pipe running down the middle of a sea ice buoy. This sea ice buoy (or ‘smartie’) will sit in a 90cm wide hole drilled through the ice. In this way the team hopes to cleanly and safely transition between under-ice and shore-based infrastructure, avoiding the problems of tidal flux and moving sea ice.
Another unique addition to the experiment is the use of two ‘mini chambers’, which can be attached to the main chambers for 24–48 hours, to run short-term experiments on individual organisms (such as a sea urchin) or to measure the effects of ocean acidification on photosynthesis. These experiments will demonstrate the potential long-term effects of ocean acidification (over 4 months) and will allow the project team to understand some of the short-term responses and acclimatization of organisms to ocean acidification.
For more information see the project website and blog.
Glenn Johnstone1 and Jonny Stark2
1antFOCE Project Manager, Australian Antarctic Division
2antFOCE Chief Investigator, Australian Antarctic Division
What is ocean acidification?
- When CO2 dissolves from the atmosphere into the ocean it increases the acidity of the ocean.
- The Southern Ocean absorbs 40% of the global ocean uptake of CO2.
- Cold water is able to absorb more CO2 than warmer water. As a result, polar waters are acidifying at twice the rate of tropical waters.
- Changes in acidity is measured on a pH scale. pH 7 is neutral, while a range between 0 and 7 is considered acidic and between 7 and 14 is alkaline. While the ocean pH is currently above pH 8, it is gradually decreasing, thereby become more acidic.
- Ocean acidification disrupts the formation of calcium carbonate (CaCO3), which is a major structural component of shells and similar hard structures made by some marine organisms, including phytoplankton and coral.
- Ocean acidification also affects the metabolic and physiological processes inside organisms including development, growth, reproduction and respiration.
- Ocean acidification will result in both winners and losers in future oceans. Some marine algae are expected to benefit from ocean acidification but most calcifying organisms are expected to suffer.
- The Intergovernmental Panel on Climate Change found that since pre-industrial times there has been an average decrease in ocean pH of 0.1 unit.
- Under a ‘business as usual’ CO2 emissions scenario, ocean pH is projected to decrease by another 0.3 to 0.4 units by 2100.
- Current atmospheric CO2 concentrations are about 416 parts per million (ppm), while ocean pH is about 8.1. By 2100 under ‘business as usual’ emissions, atmospheric CO2 is predicted to be about 936 ppm and ocean pH 7.8.
- The Antarctic research team will run their CO2-enriched chambers at 0.4 pH units below the current naturally fluctuating pH of the waters off Casey station.