Light microscopy
Both conventional compound microscopes and inverted microscopes are used to examine samples. These are used in transmitted light, Normaski differential interference contrast, phase contrast and epifluorescent modes to distinguish cellular organelles, appendages, and stained or autofluorescent cells (red autofluorescence typically denotes cellular chlorophyll).
Images are obtained using fine grained photographic film in microscope-mounted cameras or broadcast-quality video.
Living organisms are routinely examined at sea using microscopes that are isolated from the ship’s vibrations. Live material can also be examined using microscopes in a cold (0ºC) room.
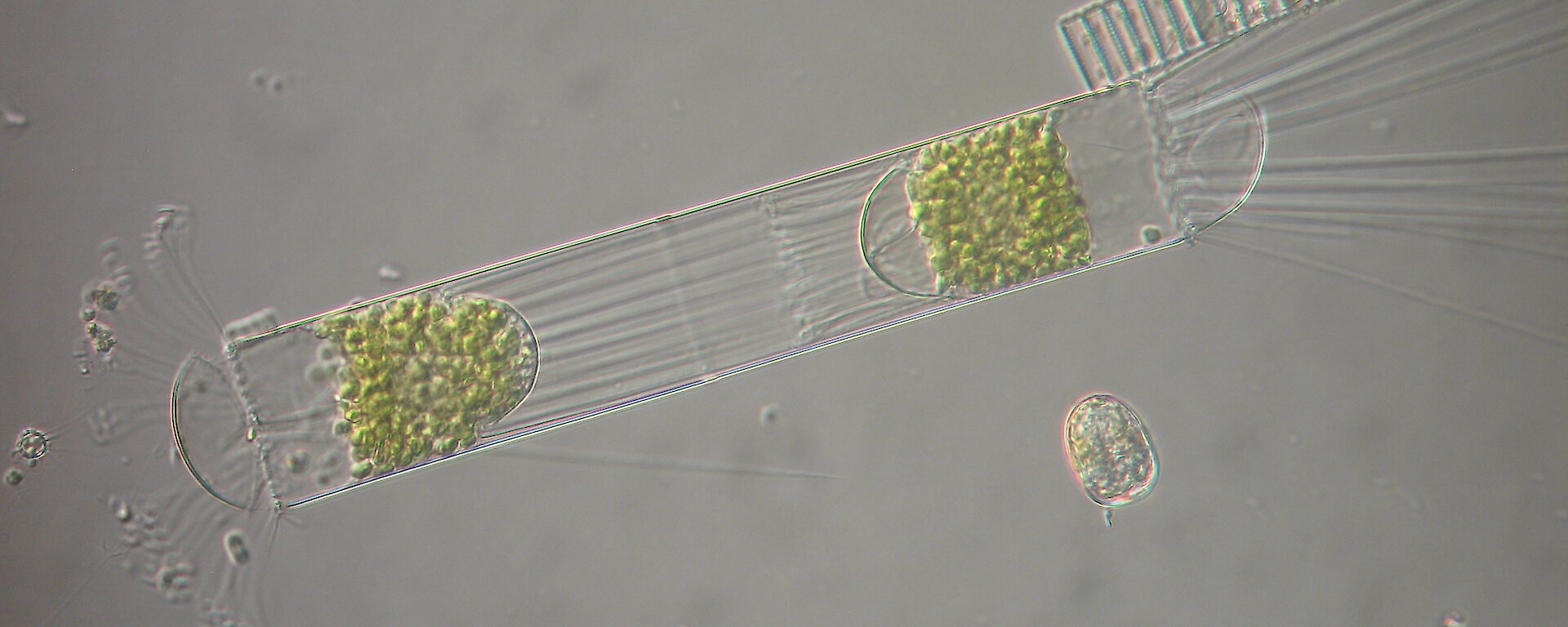
Light microscopy allows direct identification of microplankton but does not have sufficient resolution to identify most nanoplankton (especially those smaller than 10 µm). It allows cell behaviour (swimming, feeding etc.) to be studied as well as direct counts of phytoplankton. However, plankton counting is extremely time consuming, and “a task which cannot be completed without ruin of mind and body” (Haeckel, 1890).
Fluorescent stains are used to distinguish live and dead bacteria and highlight DNA and RNA of protists, bacteria and viruses.
Electron microscopy
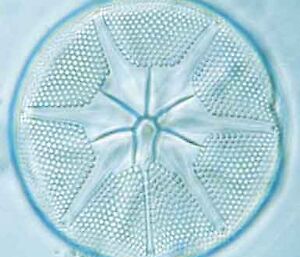
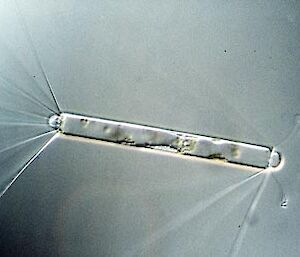
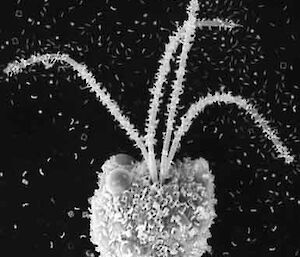
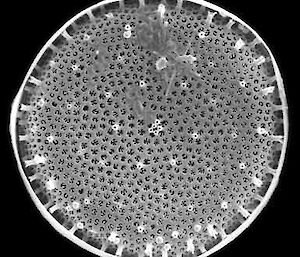
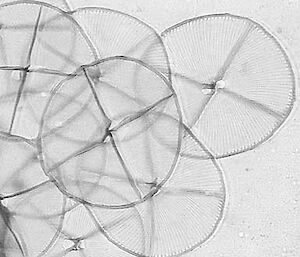
Electron microscopy is vital for the identification of small cells as well as examining details of larger cells. Much of the taxonomy of protists is based on fine structural details such as scales, flagella and surface patterning.
Scanning electron microscopy (SEM) shows the surface detail of cells.
Transmission electron microscopy (TEM) is used to examine either thin slices of material to show internal details of cells, or shadow-cast material to reveal fine surface structures of scales, flagella and other external cell components.
Flow cytometry
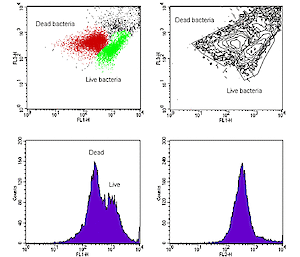
A Flow Cytometer analyses particles by passing them in single file through a laser beam. It can count up to 1000 cells per second, measuring for each one the light scattering properties (indicating size and complexity), as well as yellow-green, orange, and red fluorescence (see graphic above). Autofluorescence (from chlorophylls and other pigments) as well as the use of fluorescent stains help distinguish cell types.
The capacity to rapidly count large numbers of particles greatly increases the reliability and precision of cell concentration estimates, however the cells are not directly identified and must be examined by microscopy.
Pigment analysis
Phytoplankton, like all photosynthetic plants, use chlorophyll and carotenoid pigments to absorb light for photosynthesis. Chlorophyll a, or a derivative, is present in all types of phytoplankton and is commonly used as an indicator of the phytoplankton biomass. It enables living phytoplankton to be distinguished from zooplankton, detritus and dead phytoplankton. It is highly fluorescent and can be measured in unconcentrated seawater samples. Other chlorophylls and carotenoids are present in different types of phytoplankton. These pigments can be used as quantitative markers for particular taxonomic groups.
Fluorometry
The concentration of chlorophyll a in surface waters is continuously monitored on research voyages by measuring the fluorescence of water pumped from an intake 7 m below the ship. The temperature and salinity of this water is measured simultaneously.
Vertical profiles of chlorophyll a are measured with a fluorometer attached to the Conductivity, Temperature and Depth (CTD) instrument (see collecting field samples). This shows the vertical distribution of phytoplankton much better than discrete samples from the rosette sampler (typically collected at 15 m intervals).
Fluorescence is reduced by sunlight and the response per unit of chlorophyll varies during the day.
HPLC
HPLC (high performance liquid chromatography) is used to separate, identify and quantify the various chlorophylls and carotenoids in phytoplankton. Many of these are markers for particular taxa and can be used to estimate their contributions to the phytoplankton community.
HPLC is an excellent technique for mapping populations, since it is feasible to analyse more than 1000 samples per cruise. However it does not identify taxa directly and must be combined with microscopy to determine the key species present.
Special software (CHEMTAX) was developed collaboratively with CSIRO Marine Research to calculate the relative contributions of different groups of phytoplankton from the pigment content of field samples.
Cultures
Controlled laboratory studies of the physiology of key protist species are performed using cultures maintained at the Australian Antarctic Division. Cultures are isolated by selecting single cells from a field sample and maintained in various nutrient-enriched culture media based on seawater supplemented with various nutrients.